| Issue |
Knowl. Manag. Aquat. Ecosyst.
Number 427, 2026
Climate change impact on freshwater communities and ecosystem functioning
|
|
|---|---|---|
| Article Number | 12 | |
| Number of page(s) | 11 | |
| DOI | https://doi.org/10.1051/kmae/2026005 | |
| Published online | 03 April 2026 | |
Research Paper
Surviving drought: western pond turtles (Actinemys) persist at a pond after a historic period of low rainfall
Department of Biology, California State University, 9001 Stockdale Highway, Bakersfield, CA 93311, USA
* Corresponding author: This email address is being protected from spambots. You need JavaScript enabled to view it.
Received:
28
January
2026
Accepted:
11
March
2026
Abstract
Climate change is causing changes in weather patterns and has led to prolonged droughts. Much of western North America has experienced recent droughts, especially in California, which experienced a historic drought from 2012–2016. Drought can significantly impact aquatic turtles, especially those populations existing in ephemeral habitats. Much of the range of western pond turtles (Actinemys) has been subject to recent droughts. In 2017, after the worst of the drought had passed, I sampled a large population of Actinemys along the southern flank of the Tehachapi Mountains, California, which I first trapped in 2007 and 2010. I trapped the small pond in four of seven years between 2017 and 2023 when there was much less water than before. I caught only 15% (48/321) of the number of turtles I caught before the drought, but there were adult turtles that survived from earlier trapping, unmarked adults that were alive in 2010, young turtles, and many reproductive females. Growth rates were lower than previously, but growth rates were increasing in the last 2 years of trapping. Survivorship in 2017–2023 was similar to that in 2007/2010. Unlike many small ponds in the range of Actinemys that are ephemeral, relying on water input from the surrounding watershed, the study pond is spring-fed. Intermittent years of high rainfall (>500 mm) likely have recharged the surrounding aquifer that keep this pond with some water even in drought years, allowing the population of Actinemys at Gorman Pond to survive an extended drought period.
Key words: ecology / Emydidae / growth / pond habitat / reproduction / survivorship
© D.J. Germano, Published by EDP Sciences, 2026
 This is an Open Access article distributed under the terms of the Creative Commons Attribution License CC-BY-ND (https://creativecommons.org/licenses/by-nd/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. If you remix, transform, or build upon the material, you may not distribute the modified material.
This is an Open Access article distributed under the terms of the Creative Commons Attribution License CC-BY-ND (https://creativecommons.org/licenses/by-nd/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. If you remix, transform, or build upon the material, you may not distribute the modified material.
1 Introduction
Climate change is a worldwide phenomenon impacting all ecosystems due to human actions affecting the atmosphere (Solomon et al., 2007; Masson-Delmotte et al., 2021). Rising temperatures across the globe are changing weather patterns and increasing periods of drought in some areas. From 2000– to 2021, southwestern North America experienced the driest period in the last 1200 years (Williams et al., 2022), and the western portion of the U.S.A. and North America are projected to have higher temperatures in the 21st century, leading to more drought (Seager et al., 2007; Gutzler and Robbins, 2011). In California, the beginning of the 21st century has already brought a historic drought (Griffin and Anchukaitis, 2014; Ulrich et al., 2018), and projections are for increased periods of extreme drought (Allen and Luptowitz, 2017; Ulrich et al., 2018), although these periods may be punctuated with high rainfall events, as seen in 2016–2017 (Swain et al., 2016). The likely increased temperatures for California in the future also will produce more rain in the mountains and less snowpack (Cayan et al., 2008; Power et al., 2024), lessening summer runoff as snowmelt.
Many species of turtles have already been affected by increased temperatures and changing precipitation patterns (Butler, 2019), and populations of turtles in ephemeral habitats would likely be especially vulnerable to extended periods of drought. The two species of western pond turtles (Actinemys marmorata and Arctostaphylos pallida) are the only native freshwater aquatic turtles in California. Formerly recognized as subspecies of one species, the two taxa in the genus Actinemys were elevated to full species status by Spinks et al. (2014): the Northwestern Pond Turtle (A. marmorata) and the Southwestern Pond Turtle (A. pallida). The boundary between these species is uncertain, however, because both species and an admixture of both pallida and marmorata genes in turtles occur in the San Francisco Bay Area and along the southern edge of the Transverse Range in Los Angeles County (Spinks et al., 2010; U.S. Fish and Wildlife Service, 2023). Turtles of both species have lost much habitat in Southern California; the Central Valley, in the San Francisco Bay Area; and the Willamette Valley in Oregon (Brattstrom, 1988; Bury and Germano, 2008), and in the future, climate change could especially degrade ephemeral habitats that they use (Purcell et al., 2017; Muth et al., 2024).
In 2007 and 2010, I studied the population of western pond turtles (whether one or the other species or a genetic admixture of both) at a 1.3 ha pond near the small town of Gorman in Los Angeles County, California (Germano and Riedle, 2015). At that time, 321 turtles were marked in the two years of trapping, with a population estimate of 412 individuals. Actinemys can be long-lived, with some adults reaching an age of 50 years or more in the wild (Bury et al., 2012). Whether they can survive drought is mostly unknown. Much of the western U.S.A. and particularly California experienced drought from 2012 to 2016 (Ulrich et al., 2018), and Actinemys populations at three sites were documented to have been negatively affected (Leidy et al., 2016; Purcell et al., 2017; Lovich et al., 2017). From 2007 to 2015, rainfall near the pond near Gorman was well below average (Fig. 1), which may have dried out the aquifer feeding the pond by 2011–2012. Because of the arrival of average rainfall amounts in 2016, I returned to the pond starting in 2017 to determine if the turtles at the pond had survived this extremely dry period and, if so, how many survived and the demographic makeup of survivors.
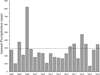 |
Fig. 1 Annual precipitation (July–June) through 2018–2019 at the closest weather station (Lebec, about 8 km straight-line distance northwest) to Gorman Pond in the Tehachapi Mountains, Los Angeles County, California, USA (https://www.weather.gov/wrh/Climate?wfo=hnx). Because the Lebec weather station ceased reporting data after 2019, rain data for 2019–2021 through 2022–2023 are from the Sandberg station, 8.5 km southeast of the pond. Year shown are the end years (e.g., 2000–2001 is shown as 2001). The dashed line is the 30-year long-term average from 1970 to 1999, excluding those rain years that had missing data during the peak rain period (October–March). |
2 Materials and methods
2.1 Study area
I sampled turtles at the pond (1063 m elevation) on the southwestern flank of the Tehachapi Mountains near the town of Gorman, Los Angeles County, California, that I had sampled before low water levels from 2011 to 2015 (Germano and Riedle, 2015). This pond has no official name, and I refer to it as Gorman Pond herein. The pond is at the base of a hillside, and a two-lane paved road is on the southern edge of the pond (34°46’57.80”N, 118°48’22.04”W, WGS84 datum). When full, the pond covers about 1.3 ha (Germano and Riedle, 2015). Gorman Pond is spring-fed and was full in 2007 when I started trapping in May, decreasing partially into August, and in 2010, it was almost full but had dried substantially by August (Fig. 2). From then until 2023, water levels in the pond never returned to full or even half full. Although I did not view the pond each year after 2010, the pond was dry by June 2014 and may have been dry all year (Fig. 2). When I started trapping again in 2017, the pond was about one third full by March-April each year thereafter (Fig. 2), and levels decreased steadily into June with no trapping possible due to low water levels by August.
 |
Fig. 2 Water levels of Gorman Pond in various years: (A) June 2007; (B) August 2010; (C) June 2014; (D) May 2019; (E) June 2020; and (F) June 2023. (All photographs are by David J. Germano, except (E) by Jessica Vannatta). |
2.2 Field methods
I captured turtles in 2017, 2019, 2020, and 2023 using both commercial nylon net traps with double funnels (model FT-FA; Nylon Net Co., Memphis, Tennessee) and homemade wire mesh traps with double funnels (see Iverson, 1979). As before, I baited traps with canned sardines, and the traps were open for either 3 or 4 d. I checked traps daily, usually starting at 0700, took turtles back to Bakersfield for processing, and released turtles the next morning back at their capture site. Because of varying water levels across years, I trapped the site four times in 2017 (28–31 March, 11–14 April, 9–12 May, 31 May to 2 June), four times in 2019 (2–5 April, 30 April to 3 May, 21–24 May, 4–7 June), three times in 2020 (28 April to 1 May, 26–29 May, 16–19 June), and three times in 2023 (15–18 May, 20–23 June, 11–13 July).
For any turtle captured for the first time during a year, I recorded mass with an electronic scale, carapace length (CL) using calipers, sex, and age following Germano (2010). I determined age using scute annuli by inspecting the costal scutes on the carapace and the abdominal scutes on the plastron (Bury and Germano, 1998; Germano and Bury, 1998). I could detect discernible scute rings at this site up to 10–12 years. I classified some turtles when first captured as older than 10 years if the shell was not well worn, yet the edges of the scutes were beveling; older than 15 years if annuli were visible but well worn; and older than 20 years if scute rings were not visible at all (Fig. 3). I found several turtles that I caught in 2017 or 2019, for which I estimated the age in 2007 and 2010, were missing one ring of growth, perhaps owing to a year when the pond did not have water. To be conservative about age, I added one year to my estimate of ages for new turtles I caught in 2017 and 2019 that were older than 2 years and still growing. Because of water being in the pond each year after 2017, I did not add a year to newly caught turtles in 2020 if <3 years old and in 2023 if <6 years old. Because some of the turtles I caught from 2017 on were first marked in 2007 or 2010 and for which I could estimate their age, I was able to determine the exact ages of some turtles >10 years. For most turtles, I defined the difference between adults and juveniles as 120 mm CL, the size at which most males develop secondary sexual characteristics in their shells and tails (Bury and Germano, 2008; Bury et al., 2012). As I did in 2007 and 2010, I individually marked turtles by notching marginal scutes with a file (Cagle, 1939; Bury and Germano, 1998).
I radiographed females in Bakersfield using a portable X-ray machine to determine if they were gravid and how many eggs were present (Gibbons and Greene, 1979; Hinton et al., 1997). At each capture period, I radiographed all females even if they previously had been captured. I used changes in the number and development of eggs (faint shell / distinct shell) as an indication of multiple clutches in a season for females with multiple captures in a year.
 |
Fig. 3 (Left) Actinemys turtle with a very worn plastron, for which the estimated age could not be determined and is assigned an age of >20 years. (Right) A turtle with well-defined annual rings for which an exact age could be estimated (photograph by David J. Germano). |
2.3 Size and growth analysis
I made comparisons of CL, weight, growth, and survivorship between turtles caught in 2017 and 2019 (17–19) and turtles caught in 2020 and 2023 (20–23) to determine if these variables differed based on time since the drought. I also compared these variables for the combined years (17–23) to those in 2007 and 2010. Although for 17–19, CL values were normally distributed and variances did not differ significantly, for all other comparisons of CL and weight, data were either not normally distributed or variances differed significantly, even after transformation. Therefore, to compare CL and weight between the sexes and time periods, I used the Kruskal–Wallis test (α = 0.05) with sex and year as groups. To determine differences between pairwise comparisons, I use the post-hoc Dunn’s test with a Bonferroni adjustment to alpha values. I only used weights of females that did not have shelled eggs. To minimize the effect of age structure on size estimates (Case, 1976), I also determined the upper quartile CLs (UQCL) and upper quartile weights (UQW) of adult turtles, and I tested for differences between years and sexes in the same manner as above. Typically, upper decile CLs and weights are used (Germano and Bury, 2009; Bury et al., 2010; Germano and Riedle, 2015; Germano, 2016), but sample sizes in each year were too low for decile fractions.
I tested for differences from a 1:1 sex ratio for 17–19 and 20–23 using chi-square analyses with Yates correction for continuity. I compared both the CL and age structures of turtles captured in 2017 and 2019 to turtles captured in 2020 and 2023 using the two-sample Kolmogorov–Smirnov Test. For all tests α = 0.05, except as noted.
As before (Germano and Riedle, 2015), I constructed growth curves by fitting age and CL data to the Richards’ growth model (Richards, 1959). The Richards’ growth model estimates three parameters using CL and age data in the general formula to solve for CL at various ages:
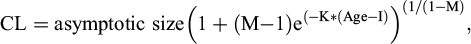 (1)
(1)
where M is the shape of the growth curve, K is the growth constant, and I is the point at which curve inflection begins. This model allows the data to construct the growth curve, not using fixed curves of other growth models.
I used continuous age estimates (Lindeman, 1997) based on a yearly period of 1 May to 30 September that could support growth. Precision of the estimate of the growth period is not critical, but estimating age to a decimal fraction of a year improves curve fit (Lindeman, 1997). Following Bradley et al. (1984), I used mean upper quartile sizes of adults as asymptotic sizes because of the high values predicted from growth data with large confidence intervals. Further, I set hatchling size to be 25–29 mm CL based on field data of recent hatchlings (Storer, 1930; Feldman, 1982; Lovich and Meyer, 2002) to anchor growth curves. I made comparisons of growth rates between sexes and between time periods using the statistic G, which represents the time required to grow from 10% to 90% of asymptotic size and is an indicator of the duration of primary growth (Bradley et al. 1984):
 (2)
(2)
The raw parameters K and M are closely linked in determining growth curves, and neither is useful for comparing growth between populations (Bradley et al., 1984). The best overall growth measure is G because it is less affected by instability of the non-linear fit than either K or M, and it produces values on an easily interpreted scale (Bradley et al., 1984); in this case, years.
I compared the growth of turtles at Gorman Pond in 17–19 to that in 20–23 and to the growth of turtles in 2007 and 2010. I compared the growth parameters and asymptotic CLs for adults irrespective of sex. I also used the growth equations to estimate the time it took for turtles to reach 120 mm CL, the size at which females can be distinguished from males based on shell characteristics, among time periods. In addition, I estimated ages to reach 80 mm and 150 mm CL to compare to ages estimated when I first trapped at the site.
2.4 Survival and population size analysis
I calculated recapture and demographic vital rates using encounter histories derived from individual encounter histories in Program MARK (White and Burnham, 1999). I calculated population size (N), apparent survival (Φ), and recapture rates (p) using open population Cormack–Jolly–Seber (CJS) and POPAN models in Program MARK (Lebreton et al., 1992; White and Burnham, 1999). I generated CJS model sets based on sex (male, female, juvenile) to test whether Φ or p was best estimated independent of sex or time, by sex or time, or a sex-time interaction. Model selection was based on Akaike Information Criterion (AICc) values, with lower values denoting greater parsimony (Burnham and Anderson, 2002).
Traditionally, encounter rates are used to calculate the probability that an individual will leave a population. If the encounter rates are reversed, then the probability of an individual entering the population can be estimated (Pradel, 1996), where Lambda (λ) = rate of individuals entering a population or cohort. Using Pradel models, λ estimates the realized growth rates of the age class from which the encounter rates were generated but is not necessarily equivalent to the growth rate of the population. Still, it provides an important metric of the life-history characteristics of a population. Pradel’s λ was estimated by Program MARK in conjunction with the CJS model described above.
3 Results
I caught 27 individual pond turtles in 2017 and 32 in 2019 (Fig. 4), representing 11 males (six new), 18 females (five new), and 9 juveniles (all new). Three juveniles caught in 2017 grew enough to be categorized as females in 2019. Other than turtles for which I could not estimate age, ages ranged from turtles in their 6th year to those in their 14th year (Fig. 4). I caught 29 individuals in 2021 and 34 individual turtles in 2023. In these two years (Fig. 4), there were 12 different males (one new), 23 different females (one new), and 6 juveniles (five new). For those turtles of which I could determine age, turtles ranged in age from their 3rd year to those in the 18th year (Fig. 4). In the four years I trapped for turtles, I captured 48 different individuals. Of these, 19 were marked in 2007/2010, and I also caught four males and six females that would have been alive before the drought.
The earliest dates I found eggs in a female were 10 May in 2017, 30 April in 2019, 28 April in 2020, and 11 July in 2023, and the percentage of females that were gravid in a year varied from 33.3% to 53.3% (Tab. 1). Over the 4 years, however, 60.0% of females (15 of 25) were found with eggs. Average clutch size varied from 5.3 to 6.0 (Tab. 1), with the preponderance (58.8%) of clutches having 5–6 eggs. There were 34 clutches when all years are combined, with a mean of 5.6 ± 0.22 (standard error) eggs per clutch. One female (161 mm CL, 20+ years old) produced a second clutch in each of the first 3 years of trapping. The smallest female with eggs (4) was 129 mm CL (8.24 years), and the youngest female with eggs (4) was 7.32 years old (134 mm CL). Clutch size increased significantly with increasing female CL (F1,28 = 5.73, P = 0.024), although only 17.0% of the variation in clutch size was explained by female CL.
The mean CLs of turtles differed significantly by sex and year (H = 9.56, df = 3, P = 0.023), although only males in 20–23 were significantly larger than males in 17–19 (Z = 2.628, P = 0.009; Tab. 2). No other comparisons of CL by sex and year differed significantly (Z = 0.438–2.554, Ps = 0.012–0.661). The overall mean CL of adults (sexes combined) in 20–23 was significantly larger than that of adults caught in 17–19 (H = 7.595, df = 1, P = 0.006). Similarly, the mean UQCL differed significantly (H = 4.482, df = 1, P = 0.034), with turtles caught in 20–23 larger than those caught in 17–19 (Tab. 2). Mean weights differed significantly among comparisons (H = 19.410, df = 3, P < 0.001), but only the mean weight of females in 20–23 was significantly heavier than that of males caught in 17–19 (Z = 4.260, P < 0.001; Tab. 2). No other comparisons of weight by sex and year differed significantly (Z = 0.893–2.550, Ps = 0.011–0.130). The overall mean weight of adults in 20–23 was significantly larger than that of adults caught in 17–19 (H = 6.465, df = 1, P = 0.011), although UQW did not differ significantly (H = 0.918, df = 1, P = 0.338) between years (Tab. 2).
The sex ratio of males to females in 17–19 (11:20 or 0.550) was not significantly different from 1:1 (X2 = 2.065, P = 0.151), nor did the ratio in 20–23 (12:23 or 0.522) differ significantly (X2 = 2.851, P = 0.091). In 2017–2019, 23.7% of turtles were juveniles (<120 mm CL), whereas in 20–23, 14.6% of turtles were juveniles (Fig. 4). The size structures between time periods differed significantly (D = 0.350, P = 0.013). No turtles were <5 years old in 17–19, although nine (30.0% of total caught) were between their 6th and 8th year of growth. In 20–23, six turtles (19.4% of total) were between their 4th and 11th year of growth, and three juveniles were <5 years old (Fig. 4). Because of past captures in 2007 and 2010, I could estimate the age of 70.0% of turtles in 17–19 and 71.0% of turtles in 20–23, and the age structures differed significantly between time periods (D = 0.430, P = 0.012) with a larger spread of ages in 20–23 (Fig. 3).
The growth model fit the data well with coefficients of determination of 0.950 for males and 0.928 for females in 17–19 and 0.937 for males and 0.936 for females in 20–23. Growth of females was not significantly different from males in 17–19 or in 20–23 (Fig. 5), but growth was significantly faster in 20–23 than in 17–19 until about age 8 years (Fig. 6). Model parameters in 17–19 for adults (irrespective of sex) were M = 2.2696, K = 0.4194, and I = 4.5795, and for 20–23 adults, they were M = −0.1130, K = 0.1818, and I = −1.4391. These values gave estimated ages that were lower to reach 80 mm CL and 120 mm Cl in 20–23 than in 17–19 although it took longer to reach 150 mm CL (Tab. 3). The 10%–90% growth parameter (G) was almost the same between time periods, taking about 11.5 years for the majority of growth to occur (Tab. 3).
The best Cormack–Jolly–Seber model of survivorship and capture probability of turtles across all years was static survivorship and recapture rate, irrespective of group (Tab. 4). Apparent yearly survival was 0.893 for males, 0.994 for females, and 0.956 for juveniles, and λ values denoted a stable population (Tab. 5). Recapture rates were high, with males at 1.00 and juveniles having the lowest rate at 0.891 (Tab. 5). Combining all groups, the population size estimate from 2017 to 2023 was 51 turtles, varying from a low of 48 (the actual number of individuals caught) to a high of 75 (Tab. 5).
 |
Fig. 4 Frequency distribution of carapace lengths (left) and ages (right) in 2017 and 2019 (top) and 2020 and 2023 (bottom) of western pond turtles (Actinemys) captured at Gorman Pond, Los Angeles County, California. Black bars are males, striped bars are females, and light grey bars are turtles for which sex could not be determined. |
The number (n), mean (±standard error), and range of eggs produced and the percentage (%) of gravid female western pond turtles (Actinemys) captured at Gorman Pond, Los Angeles, California, USA in 4 years following a severe drought. The value of n Gravid does not match n in the first 3 years because one female double clutched in each of the first 3 years.
Sample size (n), mean (±standard error, S ), range of carapace length (CL) and weight, and upper 25% CL (UQCL) and weight (UQW) of adult western pond turtles (Actinemys) captured at Gorman Pond, Los Angeles, California, USA, in 2017 and 2019, and 2020 and 2023. Carapace lengths are for the last capture, and weight is the heaviest of the turtles obtained and does not include recaptures in either time period. Weight of females excludes those that were gravid. For mean CL, males were significantly longer in 2020 and 2023 than in 2017 and 2019, and no other comparisons were significant. Both combined mean CL (all) and UQCL in 2020 and 2023 were significantly larger than for 2017 and 2019. For weight, females in 2020 and 2023 were significantly heavier than males in 2017 and 2019, and for combined weights (all), turtles were heavier in 2020 and 2023 than in 2017 and 2019, although UQW did not differ significantly between time periods
), range of carapace length (CL) and weight, and upper 25% CL (UQCL) and weight (UQW) of adult western pond turtles (Actinemys) captured at Gorman Pond, Los Angeles, California, USA, in 2017 and 2019, and 2020 and 2023. Carapace lengths are for the last capture, and weight is the heaviest of the turtles obtained and does not include recaptures in either time period. Weight of females excludes those that were gravid. For mean CL, males were significantly longer in 2020 and 2023 than in 2017 and 2019, and no other comparisons were significant. Both combined mean CL (all) and UQCL in 2020 and 2023 were significantly larger than for 2017 and 2019. For weight, females in 2020 and 2023 were significantly heavier than males in 2017 and 2019, and for combined weights (all), turtles were heavier in 2020 and 2023 than in 2017 and 2019, although UQW did not differ significantly between time periods
 |
Fig. 5 Growth curves (with 95% prediction intervals) using the Richards growth model of male (squares) and female (circles) western pond turtles (Actinemys) captured at Gorman Pond, California, in 2017 and 2019 and 2020 and 2023. |
 |
Fig. 6 Overall growth curves of western pond turtles (Actinemys) at Gorman Pond for 2017 and 2019 (Xs) compared to turtles from 2020 and 2023 (diamonds). |
Asymptotic (AS) carapace length (CL; mm) and age of western pond turtles (Actinemys) from Gorman Pond, Los Angeles County, California, USA, at three carapace lengths (mm) of turtles after a period of low rainfall (2017, 2019, 2020, 2023) and before low water levels (2007, 2010) based on Richards' growth curves for these time periods. Also shown are the times required to grow from 10–90% of asymptotic size (G) in years.
Cormack–Jolly–Seber model set analyzing the effects of sex (male, female, juvenile) and time on apparent survivorship (Φ) and recapture rates (p) of western pond turtles (Actinemys) captured at Gorman Pond, Los Angeles County, California, USA, from 2017 to 2023. Only the first five models are shown.
Apparent annual survivorship (Φ), recapture rate (p), population size (N), and lambda (λ), plus upper and lower 95% confidence intervals for western pond turtles (Actinemys) captured at Gorman Pond, Los Angeles County, California, USA, from 2017 to 2023.
3.1 Comparison of populations before and after the drought
The average clutch size for females from 2017 to 2023 was 5.6 eggs (range of values 3–9 eggs), whereas the average clutch size in 07–10 was 6.3 eggs (1–11). The percentage of females with eggs in 07–10 was 75.5% (unpublished data), and 60.0% of females were gravid during 17–23. I caught 321 individual Actinemys in 2007/2010 but only caught 48 individuals from 2017 to 2023. The largest females in 2007 averaged 159.8 mm CL and 158.9 mm CL in 2010. In 17–19, the largest females averaged 152.6 mm CL and 154.2 mm in 20–23. For males, the largest average CLs were 174.4 mm in 2007 and 173.6 mm in 2010, whereas in 17–19, the largest males averaged 149.7 mm CL and in 20–23, 156.3 mm CL. The growth curves of Actinemys differed significantly (Fig. 7) with much higher rates of growth when water levels were high (07–10) than when water levels were low. These rates of growth led to turtles reaching 80–150 mm CL and accruing the majority of their growth (10–90%) at much earlier ages in 07–10 than in 2017 to 2023 (Tab. 3). Survivorship and Pradel’s λ were similar between trapping periods, however. Estimated annual survivorship from 2017 to 2023 was 0.893 for males, 0.994 for females, and 0.956 for juveniles. In 07–10, estimated monthly survivorship was 0.989 for males, 0.994 for females, and 0.999 for juveniles (Germano and Riedle 2015), which converts to annual survivorships of 0.876 (males), 0.930 (females), and 0.988 (juveniles). Also, estimates of λ were close to 1.00 for all groups for both time periods.
 |
Fig. 7 Growth curves of western pond turtles (Actinemys) at Gorman Pond comparing growth before a period of low rainfall in 2007 and 2010 (diamonds) to growth after a period of low amounts of precipitation preceding 2017 to 2023 (squares). |
4 Discussion
If continuing climate change increases the duration of low rainfall in the range of Actinemys (either/both species), ephemeral habitats such as livestock ponds, which support many turtle populations (Tu and Trulio, 2022; Germano et al., In Press; Bury et al., In Press), could become uninhabitable. Unless turtles can find permanent water nearby in which to migrate, these populations may go extinct if ponds are dry for several years. There have already been cases of extreme drought greatly affecting Actinemys populations. In a stock pond in the foothills of the Sierra Nevada, seven of 16 radio-telemetered A. marmorata were known dead after the pond dried in late summer 2013 and did not have water again until spring 2015 (Purcell et al., 2017). By March 2015, only two telemetered turtles remained (adult male and female), and each had moved long distances to new water sources nearby: a male about 650 m and a female 2.6 km (Purcell et al., 2017). The male that moved to a new water source survived out of water for 617 days, which could have been possible because some rain fell while it was not in a water body that could have allowed for partial rehydration. At a long, narrow, shallow pond containing a population of A. pallida at the edge of the Mojave Desert, about 45 km southeast of Gorman Pond, a fire destroyed surrounding vegetation in 2013 in the middle of the drought (Lovich et al. 2017). By September 2015, the pond was completely dry, and dead and dying turtles were found. The authors estimated that the turtle population was 170 turtles in 2013 when the pond still held water but was effectively zero by late 2015. They took 32 emaciated turtles to a turtle rehabilitation center (Lovich et al., 2017). The early 2000s drought also dried the upper Amargosa Creek in the western Mojave Desert, extirpating the A. pallida population there (Muth et al., 2024).
An extended drought may not necessarily extinguish populations of Actinemys in all ephemeral habitats, however. A survey in 2017 of 16 livestock ponds in the Diablo Range of Santa Clara County, California, known to be occupied by A. marmorata before the 2012–2016 drought, found turtles extant at 12 of these ephemeral ponds (Tu and Trulio, 2022). Thus, much depends on the length of time between wetter periods and how much rain then falls. Some predictions for California are that lengthening dry periods will be punctuated with higher rainfall events than have typically occurred in the past (Swain et al., 2016; Allen and Luptowitz, 2017).
From 2000 to 2023, only in the 2004–2005 rainfall year at Gorman Pond was there a high amount of precipitation (>500 mm). Even though rainfall in 2007 and 2010 was well below average, the water level in Gorman Pond was either very high (2007) or high (2010) when I trapped the site. It is likely that the very high rain amounts in the 2004–2005 rain year charged the surrounding aquifer enough to keep filling the pond until 2011–2012. From 2006–2007 until 2015–2016 (9 years), rainfall amounts remained below average. From 2015–2016 to 2022–2023 (7 years), five of the rainfall years were above average, but not extremely high, and this likely partially recharged the aquifer feeding the pond. At least at this site fed by spring water, water levels, and likely the survival of turtles, depend on occasional high rainfall events to recharge the aquifer, which may occur despite extended drought.
Because Gorman Pond is spring-fed, turtles at this site may persist for decades, even in the face of climate change. Still, however, much depends on the timing of rainfall events. At this site, annual rainfall (July–June) amounts for 30 years from 1970 to 1999 varied widely (Fig. 8). From 1972–1973 to 1992–1993, precipitation totals near Gorman Pond exceeded 500 mm every 4–6 years. In the succeeding 7 years, high rainfall amounts (>500 mm) occurred four times (Fig. 8). I suspect that these high rainfall amounts spread out across 30 years meant that the pond near Gorman had some water throughout this time. This periodicity of high precipitation amounts may allow Actinemys to survive in Gorman Pond and other systems with an aquifer that supplies water.
The population of Actinemys at Gorman Pond after 9 years of low rainfall differed from before this event, but all indications are that turtles there constitute a functioning population. Although fewer turtles occurred at the site after the drought than before 2011, the numbers remained stable from 2017 to 2023. I found 19 turtles first marked in 2007/2010, some unmarked adults that were alive in 2010 (based on their age), young turtles, and many females that were reproductive. Overall, the growth rate was significantly lower in 2017–2023 than in 2007/2010, but growth was significantly higher in 2020/2023 than in 2017/2019, indicating that growth rates might return to pre-low water levels even if only moderate amounts of water continue at the pond. The largest female CLs were not substantially different between pre- and post-high-water levels, but the largest males in 2007/2010 had much larger CLs at that time than males after the drought. It is possible that the very large males before the drought suffered significantly higher mortality than smaller males, although larger turtles have a lower surface to volume ratio that should be an advantage for conserving body water. Within time periods, however, survivorship is about the same for turtles after the drought as before the drought. At least through 2023, the population of Actinemys at Gorman Pond survived an extended drought. It would be important for the conservation of Actinemys turtles to revisit sites after drought periods to determine the fate of populations.
 |
Fig. 8 Annual precipitation (July–June) from 1970 to 1999 near Gorman Pond in the Tehachapi Mountains, Los Angeles County, California, USA. To determine the historical rainfall patterns before 2000, I used two weather stations: the Lebec station (about 8 km straight-line distance northwest of the pond) and Sandberg (about 8.5 km southeast of the pond), each of which had missing data for some years during the period of most rain (November–March). Between the two stations, however, I had complete or almost complete precipitation totals in these years. The dashed line is at 500 mm of precipitation, indicating high rainfall years (asterisks; see the Discussion section). |
Acknowledgments
I thank Jessica Vannatta, who helped with trapping several times in 2020 and 2023. I also thank R.B. Bury and J. Lovich for reviewing an earlier draft of this paper. Work on turtles was done under a California Scientific Collecting Permit (SC-955) and a Memorandum of Understanding from the California Department of Fish and Wildlife.
References
- Allen RJ, Luptowitz R. 2017. El Niño-like teleconnection increases California precipitation in response to warming. Nat Comm 8:16055. https://doi.org/10.1038/ncomms16055. [Google Scholar]
- Bradley DW, Landry RE, Collins CT. 1984. The use of jackknife confidence intervals with the Richards curve for describing avian growth patterns. Bull South Calif Acad Sci 83: 133–147. [Google Scholar]
- Brattstrom BE. 1988. Habitat destruction in California with special reference to Clemmys marmorata: a perspective. In: De Lisle HF, Brown PR, Kaufman B, McGurty BM, eds. Proceedings of the Conference on California Herpetology. Special Publication No. 4, Southwestern Herpetologists Society. pp. 13–24. [Google Scholar]
- Burnham KP, Anderson DR. 2002. Model Selection and Multimodel Inference. Springer-Verlag. [Google Scholar]
- Bury RB, Germano DJ. 1998. Annual deposition of scute rings in the western pond turtle, Clemmys marmorata. Chelonian Conserv Biol 3: 108–109. [Google Scholar]
- Bury RB, Germano DJ. 2008. Actinemys marmorata (Baird and Girard 1952): Western Pond Turtle, Pacific Pond Turtle. In: Rhodin AGJ, Pritchard PCH, van Dijk PP, Saumure RA, Buhlman KA, Iverson JB, eds. The Conservation Biology of Freshwater Turtles and Tortoises. A Compilation Project of the IUCN/SSC Tortoise and Freshwater Turtle Specialist Group. Chelonian Research Monographs No.5. pp. 001.1–001.9 [Google Scholar]
- Bury RB, Germano DJ, Bury GW. 2010. Population structure and growth of the turtle Actinemys marmorata from the Klamath-Siskiyou ecoregion: age, not size, matters. Copeia 2010: 443–451. [Google Scholar]
- Bury RB, Ashton DT, Welsh Jr HH, et al. 2012. Synopsis of biology. In: Bury RB, Welsh Jr HH, Germano DJ, Ashton DT, eds. Western Pond Turtle: Biology, Sampling Techniques, Inventory and Monitoring, Conservation, and Management. Northwest Fauna 7. pp. 9–19. [Google Scholar]
- Bury RB, Germano DJ, Ashton DT. In Press. >Actinemys marmorata (Spinks et al. 2014) – Northwestern Pond Turtle>. CFR Species Account. [Google Scholar]
- Butler CJ. 2019. A review of the effects of climate change on chelonians. Diversity 11, 138. https://doi.org/10.3390/d11080138. [Google Scholar]
- Cagle FR. 1939. A system for marking turtles for future identification. Copeia 1939: 170–173. [Google Scholar]
- Case TJ. 1976. Body size differences between populations of the Chuckwalla, Sauromalus obesus. Ecology 57: 313–323. [Google Scholar]
- Cayan DR, Maurer EP, Dettinger MD, et al. 2008. Climate change scenarios for the California region. Clim Chang 87(Suppl 1): 21–42. [Google Scholar]
- Feldman M. 1982. Notes on reproduction in Clemmys marmorata. Herpetol. Rev. 13: 10–11. [Google Scholar]
- Germano DJ. 2010. Ecology of the western pond turtle (Actinemys marmorata) at sewage-treatment facilities in the San Joaquin Valley, California. Southwest Nat 55: 89–97. [Google Scholar]
- Germano DJ. 2016. The ecology of a robust population of Actinemys marmorata in the San Joaquin Desert of California. Copeia 104: 663–676. [Google Scholar]
- Germano DJ, Bury RB. 1998. Age determination in turtles: evidence of annual deposition of scute rings. Chelonian Conserv Biol 3: 123–132. [Google Scholar]
- Germano DJ, Bury RB. 2009. Variation in body size, growth, and population structure of Actinemys marmorata from lentic and lotic habitats in southern Oregon. J Herpetol 43: 510–520. [Google Scholar]
- Germano DJ, Riedle JD. 2015. Population structure, growth, survivorship, and reproduction of Actinemys marmorata from a high elevation site in the Tehachapi Mountains of Southern California. Herpetologica 71: 102–109. [Google Scholar]
- Germano DJ, Bury RB, Valdez-Villavicencio JH. In Press. Actinemys pallida (Spinks et al. 2014) – Southwestern Pond Turtle. CFR Species Account. [Google Scholar]
- Gibbons JW, Greene JL. 1979. X-ray photography: a technique to determine reproductive patterns of freshwater turtles. Herpetologica 35: 86–89. [Google Scholar]
- Griffin D, Anchukaitis KJ. 2014. How unusual is the 2012–2014 California drought? Geophys Res Lett 41: 9017–9023. [Google Scholar]
- Gutzler DS, Robbins TO. 2011. Climate variability and projected change in in the Western United States: regional downscaling and drought statistics. Clim Dynamics 37: 835–849. [Google Scholar]
- Hinton TG, Fledderman P, Lovich J, et al. 1997. Radiographic determination of fecundity: is the technique safe for developing turtle embryos? Chelonian Conserv Biol 2: 409–414. [Google Scholar]
- Iverson JB. 1979. Another inexpensive turtle trap. Herpetol Rev 10: 55. [Google Scholar]
- Lebreton JD, Burnham KP, Clobert J, et al. 1992. Modeling survival and testing biological hypothesis using marked animals: a unified approach with case studies. Ecol Monogr 62: 67–118. [Google Scholar]
- Leidy RA, Bogan MT, Neuhaus L, et al. 2016. Summer die-off of western pond turtles (Actinemys marmorata) along an intermittent coast range stream in central California. Southwest Nat 61: 71–74. [Google Scholar]
- Lindeman PV. 1997. Contribution toward improvement of model fit in nonlinear regression modelling of turtle growth. Herpetologica 53: 179–191. [Google Scholar]
- Lovich J, Meyer K. 2002. The western pond turtle (Clemmys marmorata) in the Mojave River, California, U.S.A.: highly adapted survivor or tenuous relict? J Zool London 256: 537–545. [Google Scholar]
- Lovich JE, Quillman M, Zitt B, et al. 2017. The effects of drought and fire in the extirpation of an abundant semi-aquatic turtle from a lacustrine environment in the southwestern USA. Knowl Manag Aquat Ecosyst 418:1–11. https//:doi.org/10.1051/kmae/2017008. [Google Scholar]
- Masson-Delmotte V, Zhai P, Pirani A, et al., eds. 2021. Climate Change 2021: The Physical Science Basis. Contribution of Working Group I to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change. Cambridge University Press. 2391 p. [Google Scholar]
- Muth D, Lovich JE, Macip-Ríos R, et al. 2024. Aspects of the demography of a relict population of southwestern pond turtles (Actinemys pallida) in a west Mojave Desert stream in California. Northeastern Nat 31(Special Issue 12): E109–E130. [Google Scholar]
- Power ME, Chandra S, Gleick P, et al. 2024. Anticipating responses to climate change and planning for resilience in California’s freshwater ecosystems. Proc Natl Acad Sci USA 121(32) https://doi.org/10.1073/pnas.2310075121. [Google Scholar]
- Pradel R. 1996. Utilization of capture-mark-recapture for the study of recruitment and population growth rate. Biometrics 52: 703–709. [Google Scholar]
- Purcell KL, McGregor EL, Calderala K. 2017. Effects of drought on western pond turtle survival and movement patterns. J Fish Wildl Manage 8: 15–27. [Google Scholar]
- Richards FJ. 1959. A flexible growth function for empirical use. J Exp Bot 10: 290–300. [Google Scholar]
- Seager R, Ting M, Held I, et al. 2007. Model projections of an imminent transition to a more arid climate in southwestern North America. Science 316: 1181–1184. [Google Scholar]
- Solomon S, Qin D, Manning M, et al., eds. 2007. Contribution of Working Group I to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change, 2007. Cambridge University Press. [Google Scholar]
- Spinks PQ, Thomson RC, Shaffer HB. 2010. Nuclear gene phylogeography reveals the historical legacy of an ancient inland sea on lineages of the western pond turtle, Emys marmorata, in California. Mol Ecol 19: 542–556. [Google Scholar]
- Spinks PQ, Thomson RC, Shaffer HB. 2014. The advantages of going large: genome-wide SNPs clarify the complex population history and systematics of the threatened western pond turtle. Mol Ecol 23: 2228–2241. [Google Scholar]
- Storer TI. 1930. Notes on the range and life-history of the Pacific fresh-water turtle, Clemmys marmorata. Univ Calif Publ Zool 32: 429–441. [Google Scholar]
- Swain DL, Lagenbrunner B, Neelin JD, et al. 2018. Increasing precipitation volatility in twenty-first-century California. Nat Clim Chang https://doi.org/10.1038/s41558-018-0140-y. [Google Scholar]
- Tu B, Trulio L. 2022. Livestock ponds as long-term habitat for southwestern pond turtles (Actinemys pallida) in rangeland landscapes. Chelonian Conserv Biol 21: 277–282. [Google Scholar]
- Ulrich PA, Xu Z, Rhoades AM, et al. 2018. California’s drought of the future: a midcentury recreation of the exceptional conditions of 2102–2017. Earth’s Future 6: 1568–1587. [Google Scholar]
- US Fish and Wildlife Service. 2023. Species status assessment report for the northwestern pond turtle (Actinemys marmorata) and southwestern pond turtle (Actinemys pallida), Version 1.1, April 2023. U.S. Fish and Wildlife Service, Ventura Fish and Wildlife Office. https://ecos.fws.gov/ServCat/DownloadFile/241273. [Google Scholar]
- White GC, Burnham KP. 1999. Program MARK: survival estimation from populations of marked individuals. Bird Study 46: S120–139. [Google Scholar]
- Williams AP, Cook BI, Smerdon JE. 2022. Rapid intensification of the emerging southwestern North American megadrought in 2020–2021. Nature Clim Chang 12: 232–234. [Google Scholar]
Cite this article as: Germano DJ. 2026. Surviving drought: western pond turtles (Actinemys) persist at a pond after a historic period of low rainfall. Knowl. Manag. Aquat. Ecosyst., 427, 12. https://doi.org/10.1051/kmae/2026005
All Tables
The number (n), mean (±standard error), and range of eggs produced and the percentage (%) of gravid female western pond turtles (Actinemys) captured at Gorman Pond, Los Angeles, California, USA in 4 years following a severe drought. The value of n Gravid does not match n in the first 3 years because one female double clutched in each of the first 3 years.
Sample size (n), mean (±standard error, S ), range of carapace length (CL) and weight, and upper 25% CL (UQCL) and weight (UQW) of adult western pond turtles (Actinemys) captured at Gorman Pond, Los Angeles, California, USA, in 2017 and 2019, and 2020 and 2023. Carapace lengths are for the last capture, and weight is the heaviest of the turtles obtained and does not include recaptures in either time period. Weight of females excludes those that were gravid. For mean CL, males were significantly longer in 2020 and 2023 than in 2017 and 2019, and no other comparisons were significant. Both combined mean CL (all) and UQCL in 2020 and 2023 were significantly larger than for 2017 and 2019. For weight, females in 2020 and 2023 were significantly heavier than males in 2017 and 2019, and for combined weights (all), turtles were heavier in 2020 and 2023 than in 2017 and 2019, although UQW did not differ significantly between time periods
), range of carapace length (CL) and weight, and upper 25% CL (UQCL) and weight (UQW) of adult western pond turtles (Actinemys) captured at Gorman Pond, Los Angeles, California, USA, in 2017 and 2019, and 2020 and 2023. Carapace lengths are for the last capture, and weight is the heaviest of the turtles obtained and does not include recaptures in either time period. Weight of females excludes those that were gravid. For mean CL, males were significantly longer in 2020 and 2023 than in 2017 and 2019, and no other comparisons were significant. Both combined mean CL (all) and UQCL in 2020 and 2023 were significantly larger than for 2017 and 2019. For weight, females in 2020 and 2023 were significantly heavier than males in 2017 and 2019, and for combined weights (all), turtles were heavier in 2020 and 2023 than in 2017 and 2019, although UQW did not differ significantly between time periods
Asymptotic (AS) carapace length (CL; mm) and age of western pond turtles (Actinemys) from Gorman Pond, Los Angeles County, California, USA, at three carapace lengths (mm) of turtles after a period of low rainfall (2017, 2019, 2020, 2023) and before low water levels (2007, 2010) based on Richards' growth curves for these time periods. Also shown are the times required to grow from 10–90% of asymptotic size (G) in years.
Cormack–Jolly–Seber model set analyzing the effects of sex (male, female, juvenile) and time on apparent survivorship (Φ) and recapture rates (p) of western pond turtles (Actinemys) captured at Gorman Pond, Los Angeles County, California, USA, from 2017 to 2023. Only the first five models are shown.
Apparent annual survivorship (Φ), recapture rate (p), population size (N), and lambda (λ), plus upper and lower 95% confidence intervals for western pond turtles (Actinemys) captured at Gorman Pond, Los Angeles County, California, USA, from 2017 to 2023.
All Figures
 |
Fig. 1 Annual precipitation (July–June) through 2018–2019 at the closest weather station (Lebec, about 8 km straight-line distance northwest) to Gorman Pond in the Tehachapi Mountains, Los Angeles County, California, USA (https://www.weather.gov/wrh/Climate?wfo=hnx). Because the Lebec weather station ceased reporting data after 2019, rain data for 2019–2021 through 2022–2023 are from the Sandberg station, 8.5 km southeast of the pond. Year shown are the end years (e.g., 2000–2001 is shown as 2001). The dashed line is the 30-year long-term average from 1970 to 1999, excluding those rain years that had missing data during the peak rain period (October–March). |
| In the text | |
 |
Fig. 2 Water levels of Gorman Pond in various years: (A) June 2007; (B) August 2010; (C) June 2014; (D) May 2019; (E) June 2020; and (F) June 2023. (All photographs are by David J. Germano, except (E) by Jessica Vannatta). |
| In the text | |
 |
Fig. 3 (Left) Actinemys turtle with a very worn plastron, for which the estimated age could not be determined and is assigned an age of >20 years. (Right) A turtle with well-defined annual rings for which an exact age could be estimated (photograph by David J. Germano). |
| In the text | |
 |
Fig. 4 Frequency distribution of carapace lengths (left) and ages (right) in 2017 and 2019 (top) and 2020 and 2023 (bottom) of western pond turtles (Actinemys) captured at Gorman Pond, Los Angeles County, California. Black bars are males, striped bars are females, and light grey bars are turtles for which sex could not be determined. |
| In the text | |
 |
Fig. 5 Growth curves (with 95% prediction intervals) using the Richards growth model of male (squares) and female (circles) western pond turtles (Actinemys) captured at Gorman Pond, California, in 2017 and 2019 and 2020 and 2023. |
| In the text | |
 |
Fig. 6 Overall growth curves of western pond turtles (Actinemys) at Gorman Pond for 2017 and 2019 (Xs) compared to turtles from 2020 and 2023 (diamonds). |
| In the text | |
 |
Fig. 7 Growth curves of western pond turtles (Actinemys) at Gorman Pond comparing growth before a period of low rainfall in 2007 and 2010 (diamonds) to growth after a period of low amounts of precipitation preceding 2017 to 2023 (squares). |
| In the text | |
 |
Fig. 8 Annual precipitation (July–June) from 1970 to 1999 near Gorman Pond in the Tehachapi Mountains, Los Angeles County, California, USA. To determine the historical rainfall patterns before 2000, I used two weather stations: the Lebec station (about 8 km straight-line distance northwest of the pond) and Sandberg (about 8.5 km southeast of the pond), each of which had missing data for some years during the period of most rain (November–March). Between the two stations, however, I had complete or almost complete precipitation totals in these years. The dashed line is at 500 mm of precipitation, indicating high rainfall years (asterisks; see the Discussion section). |
| In the text | |
Current usage metrics show cumulative count of Article Views (full-text article views including HTML views, PDF and ePub downloads, according to the available data) and Abstracts Views on Vision4Press platform.
Data correspond to usage on the plateform after 2015. The current usage metrics is available 48-96 hours after online publication and is updated daily on week days.
Initial download of the metrics may take a while.


